 The elements whose atomic masses are written in bracket ( ) are the synthetic elements and their atomic masses values represent the Atomic Mass of the most stable isotope. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. Note: The Atomic masses are represented in the Atomic mass unit (u). The atomic number of each element increases by one, reading from left to right. The atomic weight (or atomic mass) of an element tells us on average how much one atom of a given element weighs, taking into account typical proportions of. Period A horizontal row in the periodic table. 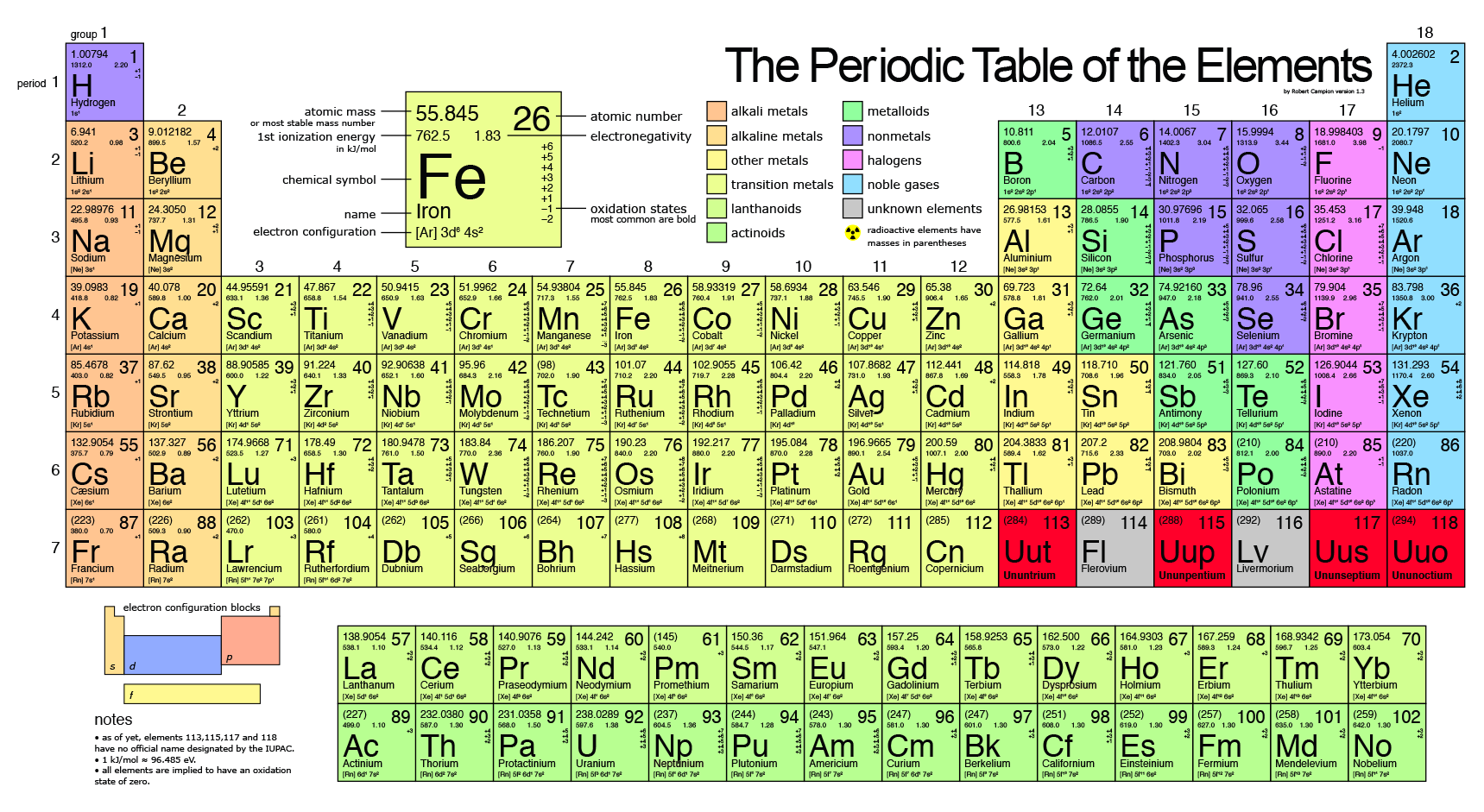
Members of a group typically have similar properties and electron configurations in their outer shell. Please note that the elements do not show their natural relation towards each other as in the Periodic system. Nitrogen 7 14.007 Glossary Group A vertical column in the periodic table. Determine the number of protons, neutrons, and electrons in an atom. The unity for atomic mass is gram per mol. The lightest chemical element is Hydrogen and the heaviest is Hassium. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). Fundamental properties of atoms including atomic number and atomic mass. Answer Click here to see a video of the solution. Calculate the average atomic mass of this element. This list contains the 118 elements of chemistry. Atomic number, atomic mass, and isotopes. An element has the following natural abundances and isotopic masses: 90.92 abundance with 19.99 amu, 0.26 abundance with 20.99 amu, and 8.82 abundance with 21.99 amu. Separation and Concentration Purification RequestĬhemical elements listed by atomic mass The elements of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects.Plant Inspection
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
